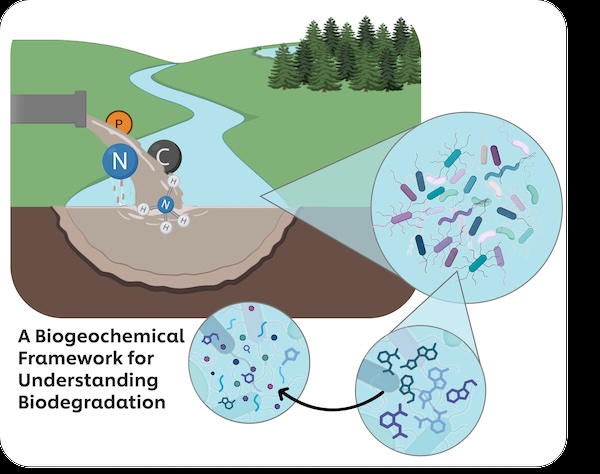
The environmental fate of synthetic polymers, including polyacrylates, polyquaterniums, and polyethylene glycols used in personal care and homecare products, remains poorly understood in natural aquatic systems. Recent European Chemicals Agency (ECHA) regulatory restrictions on polymer persistence highlight the urgent need for mechanistic understanding of biodegradation pathways. Current biodegradation assessments rely on standardised laboratory tests that may not reflect real-world environmental conditions, particularly the influence of nutrient availability on microbial degradation activity. This project addresses this knowledge gap by investigating polymer biodegradation within the context of nutrient biogeochemistry in freshwater systems.
Globally, significant volumes of untreated wastewater enter rivers and streams, creating natural gradients in nutrient availability and dissolved oxygen. These discharge zones provide ideal natural laboratories for investigating coupled nutrient-polymer dynamics. Using UK freshwater sites with documented wastewater inputs, we will characterise how nutrient conditions influence microbial communities and their capacity for polymer biodegradation. The project's primary objectives focus on testing whether nutrient availability – in particular the natural gradients created by untreated wastewater discharges - influence microbial activity associated with polymer biodegradation pathways. It will examine how elevated nutrient conditions, typical of these discharge zones, affect the breakdown of selected polymers such as those found in home and personal care products. The project will also quantify how seasonal and climate variations affect these coupled processes and develop predictive frameworks to determine how nutrient conditions found in untreated wastewater zones affect the likelihood and rate of polymer biodegradation.
We hypothesise that nutrient availability gradients created by wastewater discharge zones directly modulate microbial community composition and enzymatic activity, thereby controlling polymer biodegradation rates and pathways. Specifically, we predict that (1) Elevated nutrient conditions will alter microbial community structure, enriching for taxa with enhanced biodegradative capacity; (2) Nitrogen and phosphorus availability will correlate with expression of polymer-degrading enzymes; and (3) Seasonal variations in nutrient dynamics will create predictable patterns in biodegradation rates.
The methodology combines field measurements and laboratory analyses using isotope tracer techniques, particularly 15N, to simultaneously track nutrient transformations and polymer breakdown. Automated sampling systems equipped with multi-parameter sensors will provide continuous monitoring capabilities. Molecular techniques including amplicon sequencing will reveal community composition, while targeted quantitative PCR will examine specific degradation genes of tested polymers. Controlled microcosm studies will validate field observations and isolate specific mechanisms of polymer biodegradation. This research represents the first study to mechanistically link nutrient biogeochemistry with polymer degradation through simultaneous isotope tracking and molecular analysis. The approach constitutes a paradigm shift from treating polymer degradation as an isolated process to understanding it within broader biogeochemical cycles. Risk management strategies include laboratory microcosm studies providing controlled validation of field observations, and multiple sampling sites.
Representative Publications / References
Davenport, R., Curtis‐Jackson, P., Dalkmann, P., Davies, J., Fenner, K., Hand, L., McDonough, K., Ott, A., Ortega‐Calvo, J.J., Parsons, J.R. and Schäffer, A., 2022. Scientific concepts and methods for moving persistence assessments into the 21st century. Integrated Environmental Assessment and Management, 18(6), pp.1454-1487. DOI: 10.1002/ieam.4575
McDonough, K., Battagliarin, G., Menzies, J., Bozich, J., Bergheim, M., Hidding, B., Kastner, C., Koyuncu, B., Kreutzer, G., Leijs, H. and Parulekar, Y., 2023. Multi-laboratory evaluation of the reproducibility of polymer biodegradation assessments applying standardized and modified respirometry methods. Science of the Total Environment, 901, p.166339. DOI: 10.1016/j.scitotenv.2023.166339
Helbling, D.E., Johnson, D.R., Honti, M. and Fenner, K., 2012. Micropollutant biotransformation kinetics associate with WWTP process parameters and microbial community characteristics. Environmental science & technology, 46(19), pp.10579-10588. DOI: 10.1021/es3019012
Coll, C., Bier, R., Li, Z., Langenheder, S., Gorokhova, E. and Sobek, A., 2020. Association between aquatic micropollutant dissipation and river sediment bacterial communities. Environmental Science & Technology, 54(22), pp.14380-14392. DOI: 10.1021/acs.est.0c04393